AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Scaffold in tissue engineering ppt1/9/2024  Hydrogels usually reach their equilibrium swelling when a balance occurs between osmotic driving forces, which encourage the entrance of water or biological fluids into the hydrophilic hydrogel matrix, and the cohesive forces exerted by the polymer strands within the hydrogel. In addition, the uses of hydrogels have extended to cover a wide range of applications that include, but are not limited to, drug delivery, wound healing, ophthalmic materials and tissue engineering. Since then, the research in the field of hydrogels has expanded dramatically particularly in the last two decades. (1955–1960) 4 developed and investigated a poly(2-hydroxyethyl methacrylate)-based hydrogel for contact lens applications. The appearance of hydrogels dates back more than fifty years, when Wichterle et al. In addition, many hydrogels, such as chitosan and alginate-based hydrogels show desirable biocompatibility. In the swollen state, hydrogels are soft and rubbery, resembling to a great extent the living tissues. 
The high hydrophilicity of hydrogels is particularly due to the presence of hydrophilic moieties such as carboxyl, amide, amino, and hydroxyl groups distributed along the backbone of polymeric chains. Hydrogels can absorb huge amounts of water or biological fluids, up to several thousand %, and swell readily without dissolving. Hydrogels are three-dimensional networks composed of hydrophilic polymers crosslinked either through covalent bonds or held together via physical intramolecular and intermolecular attractions. 1 The cell-loaded scaffolds are subsequently transplanted into the patient either through direct injection with the aid of a needle or other minimally invasive delivery technique, or through implantation of the fabricated tissue at the desired site in the patient's body using surgery. The key functions of these scaffolds are to (a) deliver the seeded cells to the desired site in the patient's body, (b) encourage cell-biomaterial interactions, (c) promote cell adhesion, (d) permit adequate transport of gases, nutrients and growth factors to ensure cell survival, proliferation, and differentiation, (e) confer a negligible inflammation extent or toxicity in vivo, and (f) control the structure and function of the engineered tissue. The isolated cells are then expanded and seeded into three-dimensional scaffold that mimic the natural extracellular matrices (ECM) of the targeted tissues. The most common approach ( Figure 2) involves isolation of tissue-specific cells from the patient's small tissue biopsy and harvested in vitro. Tissues or organs can be potentially developed via a number of approaches. Also, the challenges associated with the use of hydrogel scaffolds were described.Ī schematic illustration of the four key components of tissue engineering. Special attention was given to the various design considerations for an efficient hydrogel scaffold in tissue engineering. 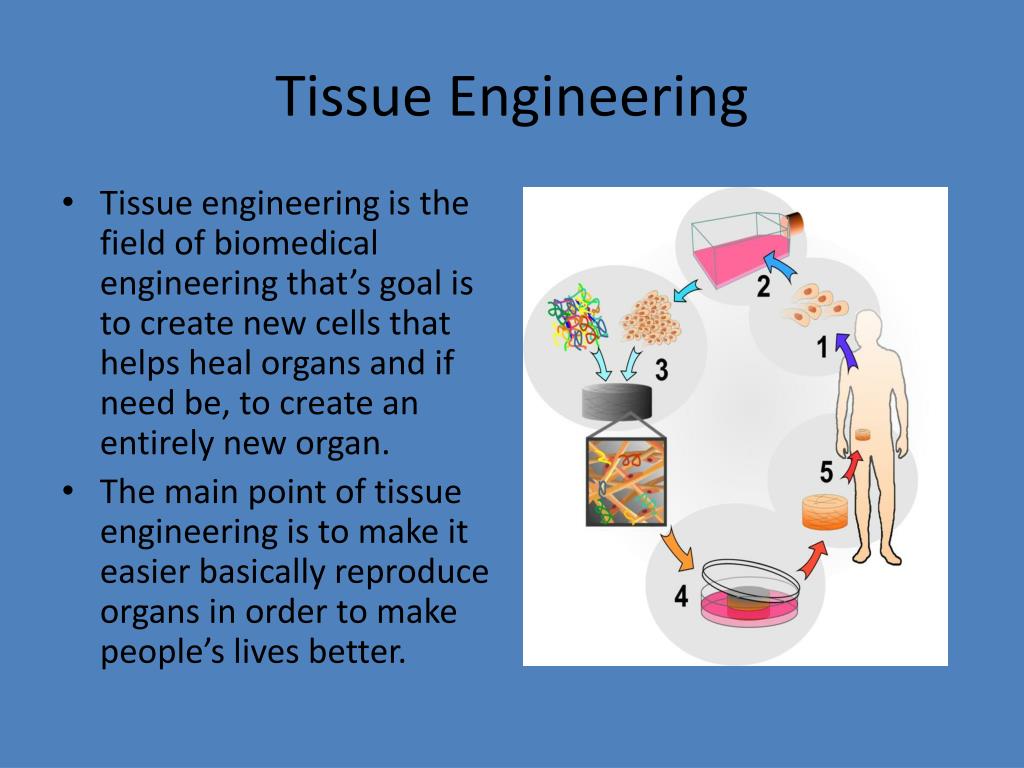
This review provides an overview of the different types of hydrogels, the approaches that can be used to fabricate hydrogel matrices with specific features and the recent applications of hydrogels in tissue engineering. More recently, the ability to control the shape, porosity, surface morphology, and size of hydrogel scaffolds has created new opportunities to overcome various challenges in tissue engineering such as vascularization, tissue architecture and simultaneous seeding of multiple cells. 
Recently, hydrogels have received a considerable interest as leading candidates for engineered tissue scaffolds due to their unique compositional and structural similarities to the natural extracellular matrix, in addition to their desirable framework for cellular proliferation and survival. Designing of biologically active scaffolds with optimal characteristics is one of the key factors for successful tissue engineering. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
